Experimental vaccination might prevent the aggressive brain tumor’s recurrence
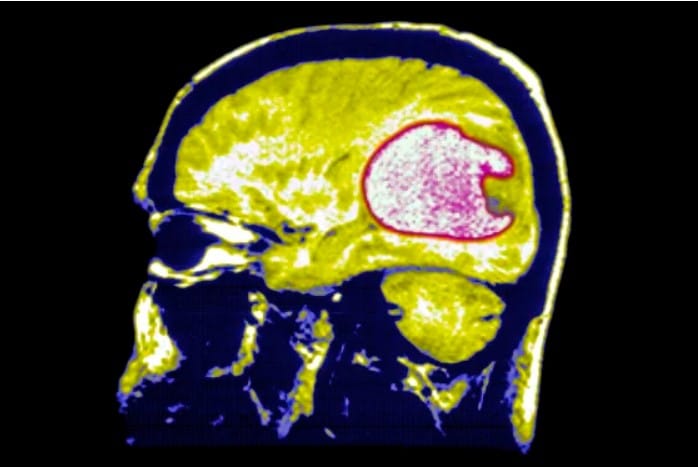
BSIP / Universal Images Group via Getty Images file
In a trial involving 63 patients, the vaccine, known as SurVaxM, was found to almost double the survival time. In a bigger experiment, researchers now want to validate the results.
In the fall of 2020, John Wishman received the grimmest brain cancer diagnosis possible: glioblastoma. He’s still traveling and having a good time two and a half years later, which is unusual for a malignancy with an average survival period of just 12 to 18 months.
Wishman, 61, of Buffalo, New York, credits this to a vaccination that is now being tested and is intended to halt the growth of the tumor. The vaccine, SurVaxM, targets a protein called survivin that is present in tumors and is so named because it is suspected to be involved in the survival of cancer cells. The idea is that by eliminating survivin, cancer cells would perish.
A vaccination that can prevent the recurrence of glioblastoma, one of the deadliest and most difficult-to-treat malignancies, seems like a pipe dream. Tom Halkin, a representative for the National Brain Tumor Society, a nonprofit organization, stated that more than 14,000 Americans received a diagnosis of a brain tumor last year. Nearly half of all malignant brain tumors are caused by it. Patients and their families are devastated by the illness; the five-year survival rate is 6.8%.
Wishman received the vaccination as a result of an extended access program, often known as compassionate use, which enables patients with life-threatening illnesses to acquire cutting-edge medications. At the Roswell Park Comprehensive Cancer Center, where the medicine is being researched, his daughter Lydia works as a nurse.
SurVaxM was reported to increase the average length of survival for those with brain cancer to 26 months in a preliminary clinical study. In an effort to verify the findings, the medication’s producer, MimiVax, located in New York, is now taking patients for a bigger experiment. The program for enhanced access is no longer offered.
Up to 270 people may join in the new experiment. The study will compare the injection to patients who receive routine treatment at more than 10 locations in the U.S. and China.
After receiving a glioblastoma diagnosis three months prior, 65-year-old Tracey Kassman joined in April 2022. She was given her first shot in the same month.
Former Buffalo attorney Kassman now receives an injection every two months. But because the experiment is double-blinded and randomized, neither Kassman nor her medical professionals are aware of whether she is receiving the vaccination or a placebo.
By the time a person is diagnosed, glioblastomas, which are aggressive malignancies, have frequently spread to other regions of the brain and spinal cord.
It is almost hard to completely remove the tumor during surgery.
According to Honggang Cui, an associate professor of chemical and biomolecular engineering at the Johns Hopkins Whiting School of Engineering, “it’s like octopus tentacles reaching into other parts of the brain.”
Radiation, chemotherapy, and surgery are frequently used as treatments, according to Cui. However, if not every cancer cell is removed, the tumor frequently returns, a condition known as recurrence.
According to Michael Ciesielski, the CEO of MimiVax, SurVaxM works by teaching the immune system to recognize and attack the cancer cells so that if they do recur, the body can eliminate them and stop the growth of a new tumor.
The method is “promising,” according to Cui. People affected by GBM may find hope in this, says the author.
According to Dr. Robert Fenstermaker, co-creator of SurVaxM and chair of the neurosurgery department at the Roswell Park Comprehensive Cancer Center, trial participants will first undergo surgery to remove as much of the tumor as possible, followed by radiation and chemotherapy using a drug called temozolomide.
“There is typically a month-long break while radiation is still working, and we like to start the vaccination during that phase because that’s when the immune system has been rejuvenated,” he added.
Four doses of the vaccine are administered over the course of two months, followed by a booster injection every two months. The vaccine is administered in the arm similarly to a flu shot or a Covid shot. For each shot, trial participants will either get the genuine vaccination or a placebo. In order to check for symptoms of advancement, participants will also get a brain scan every two months.
The development of SurVaxM is not the first effort to produce a vaccine to prevent glioblastoma from recurring. Survivin has been the target of several cancer vaccines, but none of them have, as of yet, advanced to mid- to late-stage clinical trials, according to Ciesielski.
The method is distinct from what has previously been explored, according to Dr. Alyx Porter, a neuro-oncologist at the Mayo Clinic in Phoenix.
Checkpoint inhibitors are one example of a targeted therapy that has gained popularity in recent years and improved survival in cancer patients, particularly those with breast or lung cancer. But because these medications can’t pass the blood-brain barrier, a network of blood arteries that prevents foreign substances from entering the brain, they are far less successful in treating brain tumors.